According to the law of conservation of mass, the mass of the products in a chemical reaction must equal the mass of the reactants.The law of conservation of mass states that mass in an isolated system is neither created nor destroyed by chemical reactions or physical transformations. 
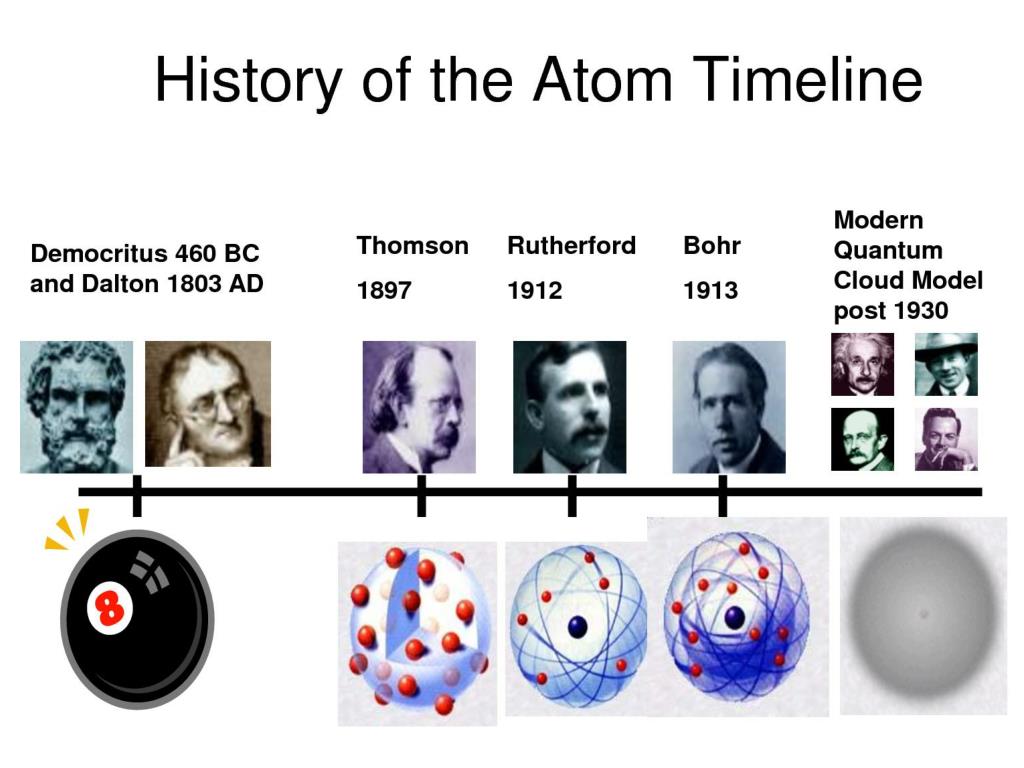
The law of conservation of mass states that mass in an isolated system is neither created nor destroyed. The video also covers the work of Dalton, Thompson, Rutherford, Niels Bohr, and Schrödinger. It starts with Democritus and Leucippus, the first philosophers to discuss atoms. “Models of the Atom Timeline” – YouTube: This video is about the different ways that scientists have pictured the atoms over the years. 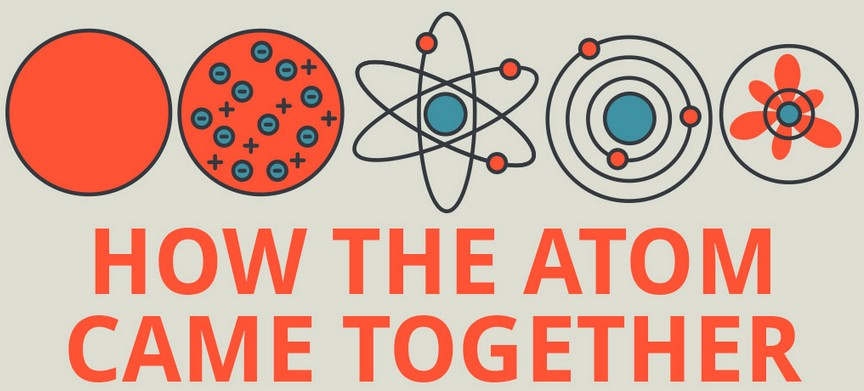
Like the early philosophers, Dalton’s theories were not popularly accepted for much of the 19th century, but his ideas have since been accepted, with amendments addressing subatomic particles and the interconversion of energy and mass. Chemical reactions may result in their rearrangement but not their creation or destruction.ĭalton also outlined a law of multiple proportions, which described how reactants will combine in set ratios.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
